IMPLASPIN

- Accelerated osseointegration
- Excellent primary stability
- High-strength
The function of the osseoconductive spinal intervertebral disc is to prevent instability of the affected motion segment of the lumbar or cervical spine.
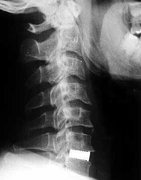
Osteoconductive implants, developed by the LASAK company, represent a new generation of biomaterials to replace bone tissue. The main characteristics of these implants are the excellent mechanical and significant osteoconductive properties of their surface, which enable faster and better healing of the implant. The implant is manufactured from pure titanium with a chemically treated surface or from PEEK-OPTIMA™ HA Enhanced with a roughened osteoconductive surface. The osteoconductive surface of the implant enables faster osseointegration without a soft tissue layer and helps to create a fusion between the implant and the bone. This strong fusion between the implant and the bone tissue ensures uniform distribution of stresses on the bone interface and loaded implant, thus preventing local overloading of the bone and subsequent bone resorption.
Indication
- Degenerative diseases of lumbar and cervical intervertebral discs
- Spinal cervical injuries
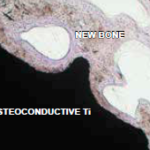
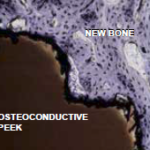
Osteoconductivity
The microstructure of an osteoconductive titanium assists the osteoblasts and osteocytes in the creation of new bone. The osteoconductive properties of the bioactive surface significantly enhance the creation of fusion between the bone and the implant.
Implants with an osteoconductive surface, compared to implants of pure titanium or PEEK show greater tolerance to unfavorable conditions for healing, such as a gap between the bone and the implant or primary implant instability.
Histological cross-sections of an implant with an osteoconductive surface and newly formed bone tissue 10 weeks after implantation. (Original magnification 200x, stained with toluidine blue.)
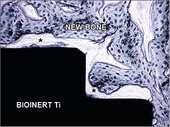
Histological cross-section of a machined surafce titanium implant and newly formed bone tissue 6 weeks after implantation, (* soft tissue). (Original magnification 200x, stained with toluidine blue.)
Histological cross-section of an implant from PEEK-OPTIMA™ HA Enhanced with an osteoconductive surface and newly formed bone tissue 6 weeks after implantation. (Original magnification 200x, stained with toluidine blue.)
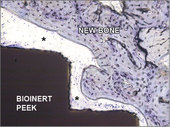
Histological cross-section of an implant PEEK-OPTIMA™ HA Enhanced with a machined surface and newly formed bone tissue 6 weeks after implantation, (* soft tissue). (Original magnification 200x, stained with toluidine blue.)
New titanium and PEEK implants with osteoconductive properties showing immediate connection with bone tissue. The healing of the implant with bioinert machined surface is almost exclusively through tissue fibers (*).
IMPLASPIN cervical intervertebral implants
IMPLASPIN lumbar intervertebral implants

INVIBIO™ and PEEK-OPTIMA™ are trade marks of Victrex plc or its group companies. All Rights Reserved.
Download
- LASAK – Capability brochure (1.09 MB)
- IMPLASPIN – product information (429.35 KB)